Member Spotlight
Dr. Peter Rahko is a faculty member in the Division of Cardiovascular Medicine within the Department of Medicine. Dr. Rahko is the Director of the Adult Echocardiography Laboratory and is active in clinical research, particularly regarding innovative new medical therapies for end-stage heart failure and is involved in designing research projects utilizing all aspects of echocardiography. Since 2003, he served as the Chair of the Minimal Risk IRB. He served in this leadership role for the lifetime of the committee until it was merged with the ED/SBS IRB in June 2021 to become the new Minimal Risk Research IRB. Now, Dr. Rahko is bringing his 18 years of IRB experience to a new leadership role as Chair of the Health Sciences IRB. Welcome to the HS IRB, Dr. Rahko!
Call for Volunteers!
We are looking for 1-2 members from each HS and MRR panel who would be willing to act as backup when the Chair is out and coverage for that roll is needed. Training would be provided and as Chairs/vice-chairs will always first try to switch meetings with another Chair/vice-chair when coverage is needed, we hope that the need to rely on backups will not be frequent. If you are interested in serving in this role, please email Catherine Rogers, csrogers@wisc.edu.
Toolkit Transition Continues! Some Reminders:
- Using HRP-314 During Meetings
Primary reviewers for initial reviews should use the “Criteria for Approval” worksheet (HRP-314) as part of their pre-meeting preparation when reviewing agenda items. During the meeting, we will display this document on the shared screen so that board members can reference the 111 criteria (section 2), when discussing whether all the required criteria have been met. For more information about the 111 criteria, please see the Continuing Education section, below.
- Reviewing All Agenda Items
IRB members are expected to review all items on an agenda, even if not assigned as the primary or secondary reviewer, in enough depth to participate in discussion about the item and to help ensure the committee reviews the submission in compliance with applicable regulatory (State and federal), institutional, and ethical requirements and standards.
Both reviewers assigned to an item should be prepared to provide the summary of an item in the case that the primary reviewer is not in attendance when the item is discussed.
- Staff Completing ARROW Checklists
For now, staff reviewers will identify and prepare all necessary checklists in ARROW. IRB members should review these in ARROW by clicking on the most recent prepared checklist under the history tab. These will be displayed during the meeting for full discussion and vote. If you see something in a checklist that seems incorrect, to avoid confusion, please do not complete a new checklist; instead, contact the staff reviewer by email or raise for discussion during the meeting.
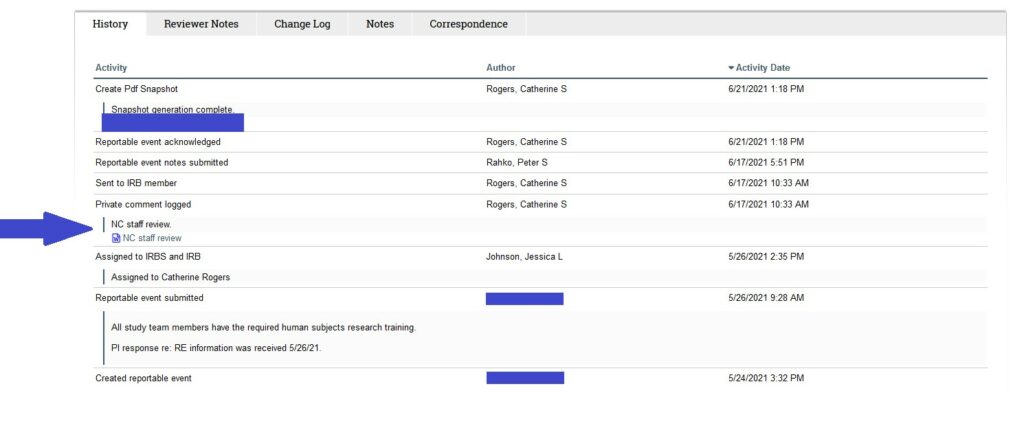
- Reportable Event Staff Reviews
Reportable Event (RE) staff reviews will be uploaded to the RE workspace as a private comment and will NOT be emailed out separately. While IRB staff no longer prepare staff reviews for most agenda items, reportable events are the singular exception. Because there are not Toolkit checklists for REs, staff reviews will continue to be prepared for the time being and will be uploaded to the RE workspace. These will be accessible in the History of the RE.
In the News
From the PRIM&R Blog: “IRBs as compliance bureaucracy? A review of Regulating Human Research” — A review of a book published immediately prior to the COVID-19 pandemic, the blog post gives a brief history of how and why IRBs were created, why some IRBs were shut down in the late 1990s, the following period of “hyper-compliance” and professionalization of IRB administration, and subsequent pushback and increased flexibility. The post raises ideas that will intrigue both new and seasoned IRB members.
Continuing Education – What are the 111 criteria?
The 111 criteria are the core regulatory criteria that an IRB must determine have been met for the approval of any non-exempt research study involving human subjects and are based on the basic ethical principles described in the Belmont Report, first written by the National Commission for the Protection of Human Subjects of Biomedical and Behavioral Research. These principles consist of the following: (a.) Beneficence, involving obligations to act for the benefit of subjects and prevention of harm. (b.) Respect for person, involving the recognition of a person as an autonomous individual with the right and capacity to make their own decisions. (c.) Justice, involving the use of non-exploitative and fairly administered procedures, fair distribution of costs and benefits to potential subjects, and appropriately justified selection of subjects.
The 111 criteria listed in HRP-314 address topics such as the minimization of risks to subjects, acceptability of the risks to subjects in relation to potential direct benefits or social/scientific benefits, equitable selection of subjects (e.g., appropriately justified subject population, enrolling the least vulnerable populations given research goals), appropriate consent process and documentation, adequate monitoring of study data to minimize risks to subjects, and appropriate provisions to protect subject privacy and maintain confidentiality.